Researchers from the University of Nebraska-Lincoln, Harvard Medical School and MIT have designed a smart bandage that could eventually heal chronic wounds or battlefield injuries with every fiber of its being.

The bandage consists of electrically conductive fibers coated in a gel that can be individually loaded with infection-fighting antibiotics, tissue-regenerating growth factors, painkillers or other medications.
A microcontroller no larger than a postage stamp, which could be triggered by a smartphone or other wireless device, sends small amounts of voltage through a chosen fiber. That voltage heats the fiber and its hydrogel, releasing whatever cargo it contains.
A single bandage could accommodate multiple medications tailored to a specific type of wound, the researchers said, while offering the ability to precisely control the dose and delivery schedule of those medications. That combination of customization and control could substantially improve or accelerate the healing process, said Ali Tamayol, assistant professor of mechanical and materials engineering at Nebraska.
“This is the first bandage that is capable of dose-dependent drug release,” Tamayol said. “You can release multiple drugs with different release profiles. That’s a big advantage in comparison with other systems. What we did here was come up with a strategy for building a bandage from the bottom up.
“This is a platform that can be applied to many different areas of biomedical engineering and medicine.”
The team envisions its smart bandage being used initially to treat chronic skin wounds that stem from diabetes. More than 25 million Americans – and more than 25 percent of U.S. adults 65 and older – could suffer from such wounds. The Centers for Disease Control and Prevention has estimated that diabetes cases will double or triple by the year 2050.

“The medical cost associated with these types of wounds is tremendous,” Tamayol said. “So there is a big need to find solutions for that.”
Those wounded in combat might also benefit from the bandage’s versatility and customizability, Tamayol said, whether to stimulate faster healing of bullet and shrapnel wounds or prevent the onset of infection in remote environments.
“Soldiers on the battlefield may be suffering from a number of different injuries or infections,” he said. “They might be dealing with a number of different pathogens. Imagine that you have a variable patch that has antidotes or drugs targeted toward specific hazards in the environment.”
Bandage aid
Existing bandages range from basic dry patches to more advanced designs that can passively release an embedded medication over time. To evaluate the potential advantages of their smart bandage, Tamayol and his colleagues at Harvard ran a series of experiments.
In one, the researchers applied a smart bandage loaded with growth factor to wounded mice. When compared with a dry bandage, the team’s version regrew three times as much of the blood-rich tissue critical to the healing process.
Another experiment showed that an antibiotic-loaded version of the bandage could eradicate infection-causing bacteria. Collectively, Tamayol said, the experiments also demonstrated that the heat needed to release the medications did not affect their potency.
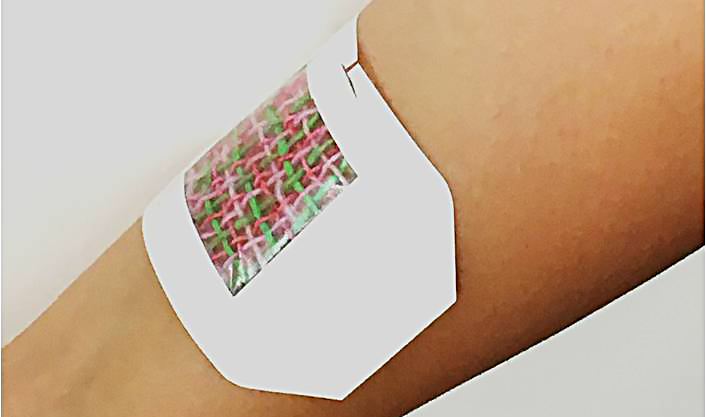
Though the researchers have patented their design, it will need to undergo further animal and then human testing before going to market. That could take several years, though the fact that most of the design’s components are already approved by the Food and Drug Administration should streamline the process, Tamayol said.
In the meantime, he said, the researchers are also working to incorporate thread-based sensors that can measure glucose, pH and other health-related indicators of skin tissue. Integrating that capability would allow the team to create a bandage that could autonomously deliver proper treatments.
The authors detailed their design and findings in the journal Advanced Functional Materials. Tamayol authored the study with Harvard’s Ali Khademhosseini, Pooria Mostafalu, Gita Kiaee, Giorgio Giatsidis, Akbar Khalilpour, Mahboobeh Nabavinia, Mehmet Dokmeci and Dennis Orgill, along with Sameer Sonkusale of Tufts University.
The researchers received support from the National Science Foundation, the Office of Naval Research and the National Institutes of Health under grants HL092836, DE019024, EB012597, AR057837, DE021468, HL099073, and EB008392.