· 4 min read
Study details formation of nitrous acid in atmosphere

The cookbook of atmospheric chemistry already contained recipes for nitrous acid, a molecular precursor to gases that both cleanse and pollute Earth’s atmosphere.
What that cookbook had lacked since the 1970s, when instrument-laden rockets and balloons first detected nitrous acid in the atmosphere, was a description of how its ingredients mixed in the highly charged layer known as the ionosphere.
A UNL-led team of chemists has finally identified these dynamics through a series of quantum mechanics-based simulations detailed April 11 in the Proceedings of the National Academy of Sciences.
The ionosphere, which stretches from about 40 miles to more than 600 miles above Earth’s surface, houses positively charged atoms and the negatively charged electrons that solar radiation regularly knocks loose from those atoms. The new study specifically describes how these charged atoms and molecules combine to form nitrous acid in the ionosphere’s lowest-lying 15 miles, which atmospheric scientists call the “D region.”
“The more we can understand how certain chemical entities are produced, that helps us to understand how our activity could be better managed in order to minimize the production of certain entities that may have environmental and health consequences,” said co-author Joseph Francisco, UNL’s Cordes Chair of Chemistry.
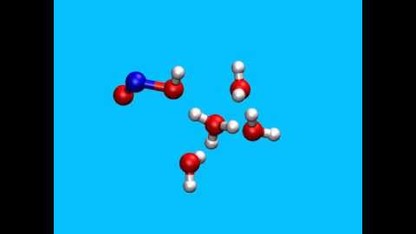
Previous research had collected the short list of necessary ingredients: a positively charged molecule consisting of a nitrogen atom triply bonded with an oxygen atom – collectively called a nitrosonium ion – plus clusters of four or five water molecules.
The UNL team’s simulations show that certain low-energy variations of those water clusters can transition into higher-energy configurations, allowing them to combine with a nitrosonium ion and, ultimately, form a nitrous acid molecule with the chemical formula HONO.
Among a series of clusters containing four water molecules, the researchers identified one particular configuration that facilitates the proper sequence of bond breakage, formation and proton transference needed to create HONO. They also found that the extra water molecule featured in five-molecule clusters helps propel even faster nitrous acid formation, though both the four- and five-molecule dynamics occur on the scale of picoseconds. One picosecond compares to a second as a second compares to 31,710 years.
Co-author Xiao Cheng Zeng, an Ameritas University Professor of chemistry, said researchers had previously detected HONO formation in the laboratory at temperatures of minus 450 degrees Fahrenheit. UNL’s simulations allowed the team to examine HONO dynamics between minus 100 and minus 60 degrees Fahrenheit, Zeng said, approximating the temperature range of the ionosphere’s D region.
“It’s like when North Carolina and Villanova played,” Zeng said, referring to the dramatic conclusion of the recent NCAA Men’s Basketball Tournament. “We knew the final score and we knew which teams played, but we wanted to see a video clip of that final 10 seconds.
“We wanted to see the dynamics at a realistic temperature of the ionosphere – make a video like that last 10 seconds of March Madness – and show that to people. That was the goal.”
Zeng credited Francisco, who also serves as dean of UNL’s College of Arts and Sciences, with stirring his interest in the chemistry unfolding in the Earth’s upper atmosphere.
“I have been studying nanoscience, water and ice for many, many years, but I’d never really looked at atmospheric science,” Zeng said. “Joe’s the one who opened up this research direction for the university and got me really interested in that direction.
“I used to look at the nanoscale – which is one-billionth of a meter – and now I’m looking up at the big sky. So Joe has been very inspirational.”
Zeng and Francisco authored the PNAS study with Rongxing He, a former UNL visiting scholar who heads the College of Chemistry and Chemical Engineering at Southwest University in China; doctoral candidate Lei Li, a 2016 recipient of UNL’s Outstanding Graduate Research Assistant Award; graduate student Jie Zhong; and postdoctoral researcher Chongqin Zhu.