· 5 min read
Researchers build molecule to suppress stroke-related enzyme

Research teams separated by 14 hours and 9,000 miles have collaborated to advance prospective treatment for the world’s second-leading cause of death.
University of Nebraska-Lincoln chemists partnered with medical researchers from the National University of Singapore to develop a molecule that can inhibit an enzyme linked with the onset of stroke.
Most strokes occur when a disruption of blood flow prevents oxygen and glucose from reaching brain tissue, ultimately killing neurons and other cells. The team found that its molecule, known as 6S, reduced the death of brain tissue by as much as 66 percent when administered to the cerebrum of a rat that had recently suffered a stroke.
It also appeared to reduce the inflammation that typically accompanies stroke, which the World Health Organization has estimated kills more than 6 million people annually.
“The fact that this inhibitor remained effective when given as post-stroke treatment … is encouraging, as this is the norm in the treatment of acute stroke,” the researchers reported in a March 9 study published by the journal ACS Central Science.
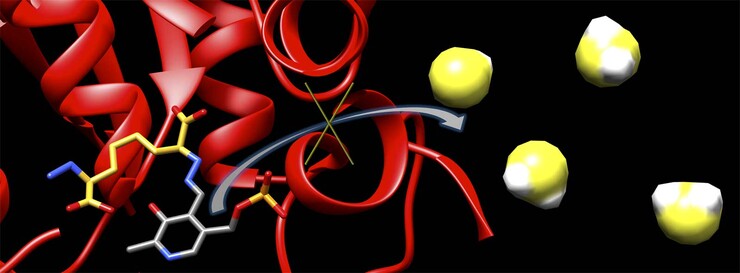
The inhibitor works by binding to cystathionine beta-synthase, or CBS – an enzyme that normally helps regulate cellular function but can also trigger production of toxic levels of hydrogen sulfide in the brain. Though hydrogen sulfide is an important signaling molecule at normal concentrations, stroke patients exhibit elevated concentrations believed to initiate the brain damage they often suffer.
Chemist David Berkowitz and his UNL colleagues modeled their inhibitor on a naturally occurring molecule produced by the CBS enzyme, tailoring the molecule’s structure to improve its performance. By swapping out functional groups of atoms known as amines with hydrazines, the team ultimately increased the inhibitor’s binding time from less than a second to hours.
“We wanted a compound that would bind well, specifically to this enzyme,” said Berkowitz, a Willa Cather Professor of chemistry. “But we also wanted one that could be synthesized easily. Those are two very different considerations.”
Berkowitz and his colleagues achieved the latter goal, in part, by plucking out the molecule’s carbon-sulfur bond and replacing it with a double bond. Slicing that double bond gave the researchers two identical halves of the molecule. With the assistance of a Nobel Prize-winning technique called cross-metathesis, the team was then able to “synthesize two halves of the molecule for the price of one,” Berkowitz said.
To test the effectiveness of the 6S molecule in treating stroke, Berkowitz and fellow UNL chemist Christopher McCune reached out to Peter Wong, professor of pharmacology at the National University of Singapore.
“We started researching this and came upon Peter’s work pretty quickly,” Berkowitz said. “We saw that he was one of the protagonists, one of the guys who is on the leading edge of understanding how (hydrogen sulfide) signaling works.”
Though the research teams have never actually met in person, Berkowitz said videoconferencing and a steady stream of emails have helped overcome the barriers of time and distance. In the process, he said, each team has developed a profound appreciation for the other’s work.
“Peter ended up latching onto the chemistry more than we did, and we ended up latching onto the biology,” Berkowitz said. “It’s actually been really fun. These are two kinds of science that are pretty far apart, and that’s probably the most exciting thing about this: the interdisciplinary nature.”
Because the 6S inhibitor has demonstrated its effects in cell cultures and the brain tissue of rats, Berkowitz cautioned that it represents just an initial step toward developing a stroke-treating drug for humans. However, he said the proof-of-principle experiments effectively illustrate the concept’s promise.
Berkowitz also expressed optimism that the synthesis method detailed in the study could streamline the more general production of enzyme-targeting inhibitors.
“We started out with a very fundamental-science perspective on understanding the chemistry of this whole class of vitamin B6-dependent enzymes,” he said. “We’re in a good place now, because that science has allowed us to make these inhibitors and many others. We’re now working on several enzymes that may represent important targets for translation of the basic inhibitor chemistry into truly therapeutic goals.”
Berkowitz, McCune and Wong co-authored the ACS Central Science study with Matthew Beio, UNL graduate student in chemistry; Weijun Shen, who earned a doctorate at UNL; Woo Jin Chung, a former UNL postdoctoral researcher; Laura Szczesniak, graduate student at SUNY Upstate Medical University; the National University of Singapore’s Su Jing Chang and Shu Qing Koh; and the National Neuroscience Institute’s Chou Chai.
Research and facilities that contributed to the study were partly funded by the American Heart Association, the National Science Foundation and the National Institutes of Health.